Discount Products: Look through products available for a 25% – 50% discount in 2020. The items provided on a first come, first serve basis. View Discounted Products
Thermo Scientific Hot Melt Extrusion
Overcome drug formulation challenges
Pharmaceutical manufacturers are using conical and twin-screw extruders to mix drug molecules with bioactive polymers in situations where drug ingredients are poorly soluble or unstable during processing. Extruders are also useful in preparing enteric dosages, sustained release dosages, in taste-masking, and to create forms such as films.
Introducing the Thermo Scientific Hot Melt Extrusion
Overcome drug formulation challenges
Pharmaceutical manufacturers are using conical and twin-screw extruders to mix drug molecules with bioactive polymers in situations where drug ingredients are poorly soluble or unstable during processing. Extruders are also useful in preparing enteric dosages, sustained release dosages, in taste-masking, and to create forms such as films.

Key Features
Polymers
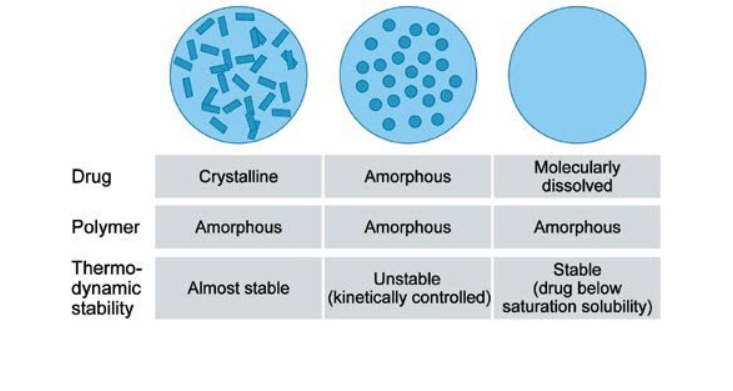
The choice of an adequate polymer as a matrix to form stable solid solutions is crucial in HME. Polymers with a high solubilization capacity are particularly suitable because they can dissolve large quantities of drugs. Polymers for hot melt extrusion experiments are based on different monomers and chemical structures, such as homopolymers, copolymers, amphiphilic copolymers as well as solubilizers and plasticizers. Consideration for each material must take into account the polymer’s solubility in a solvent, which can vary from high lipophilicity to high hydrophilicity. A suitable solvent need to be selected for the preparation of solid solutions by spray drying, where polymer and drug must be dissolved in a volatile solvent.
Polymers for hot-melt extrusion must exhibit thermoplastic characteristics and must be thermally stable at the proper extrusion temperature. In developing an HME drug system, the glass transition and melting temperatures are critical factors. The extrudability of a polymer is mainly determined by Tg or Tm and melt viscosity. Most polymers demonstrate thixotropic behavior where the viscosity reduces as a function of increasing shear stress.
Active pharmaceutical ingredients
Present drug discovery methodology produces candidate drugs of increasing molecular size and lipophilicity, which results in poor solubility and permeability. Less than 10% of new drug candidates demonstrate both high solubility and permeability, and 30–40% of the drugs that appear on the World Health Organization Essential Drug List were reported to be poorly water-soluble or lipophilic. HME disperses the drug in the matrix at a molecular level by forming a solid solution.
HME is available for a number of applications, including amorphous solid dispersion, controlled release systems, polymorph generation, novel cocrystallisation (solvent-free continuous cocrystallisation), porous polymeric systems and Process Analytical Technology (PAT).
Processing parameters
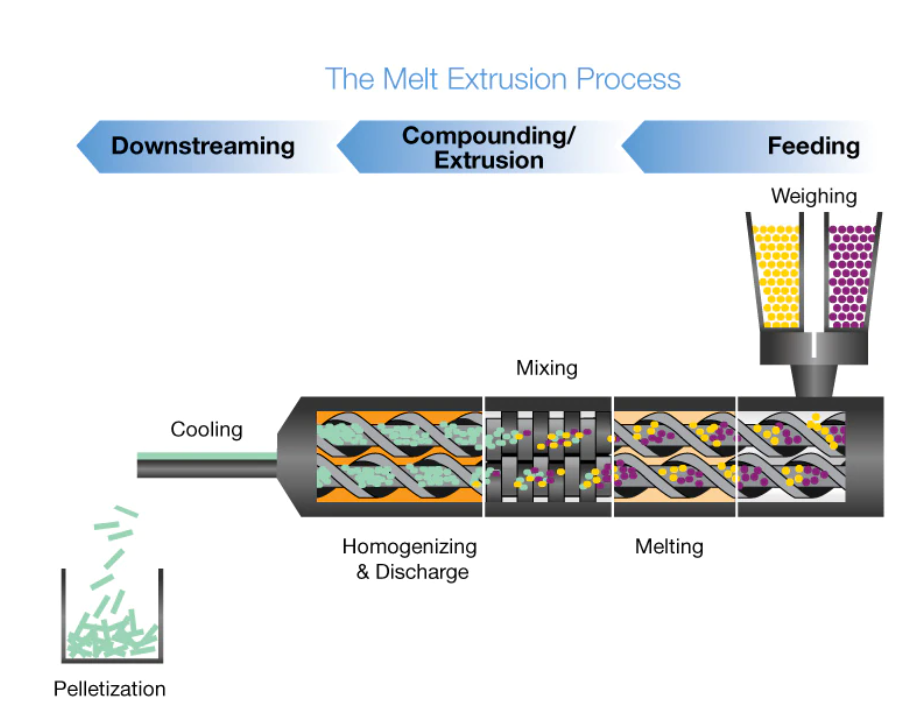
In the hot melt extrusion process, the API and the excipients are fed into the extruder. All components are sheared, heated, plastified, mixed and dispersed, and finally shaped by pressing them through a die opening. Developing a HME manufacturing project requires control of several processing parameters that affect residence time distribution, and specific mechanical energy consumption (SMEC) must be considered such as temperature of the melt at the extruder die, pressure at the die, and torque.
Key Benefits
Why hot melt extrusion?
Hot melt extrusion (HME) is the processing of polymeric materials above their glass transition temperature (Tg) in order to effect molecular level mixing of thermoplastic binders and/or polymers and active compounds. Used in several industries, HME is a combination of melting and mechanical energy to improve continuous processing for reproducible analysis of materials, dust reduction and online monitoring.
In pharmaceutical manufacturing, HME is used to disperse APIs in a matrix at the molecular level, thus forming solid solutions. This enables drug delivery systems for poorly soluble drugs or specialized drug forms such as films for transdermal patches.
Trust the Experts at Spectra Research Corporation
Spectra Research Corporation (SRC) offers a range of innovative high-quality scientific products and laboratory services to industrial and scientific markets throughout Canada.











