Sol-gel is the abbreviation of solution-gelation. Sol-gel synthesis is used in materials science to produce solids (metal oxides) from small molecules (typically metal alkoxides) and is similar to a polymerization reaction. A macromolecular network of oxides is fabricated during the hydrolysis and condensation of molecular precursors. It involves the conversion of the precursors into a colloidal solution that is converted into a gel of either discrete particles or network polymers.
Then the remaining liquid phase (water and solvent) is dried and a thermal treatment is often necessary to improve the characteristics of the final material. Dopants like organic dyes or rare earth elements can be added to the precursor in the solution and thus be dispersed in the material. The sol-gel process is considered as a cheap and low-temperature alternative to other synthesis techniques involving materials melting.
The final materials can be applied to optics, electronics, energy, gas sensors, biomaterials…
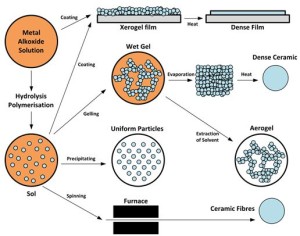
- Figure 1 – Schematic representation of the different stages and routes of the sol-gel technology (from Wikipedia)
SIMULTANEOUS THERMAL ANALYSIS
Thermogravimetry coupled to Differential Thermal analysis or Differential Scanning Calorimetry is a long time proven method to study thermal processes. It is mostly applied in the field of sol-gel synthesis to determine the temperature ranges of interest during the drying and thermal treatment phases.
A typical TGA thermogram corresponding to the heating of a gel under controlled rate exhibits a multiple steps mass loss corresponding to the evaporation of water and organic solvents. The DTA or DSC signal will exhibit endotherms due to evaporation or exotherms when the evaporating solvents decompose or oxidize. At higher temperature an exotherm not linked to any mass change is observed, corresponding to the crystallization of the synthesized amorphous material. Depending on the desired final product, this crystallization temperature may be a limit to overcome or not.

