Oxford Instruments Asylum Research announces its new application note describing atomic force microscopy (AFM) tools for nanoelectrical characterization. The application note discusses the most recent nanoelectrical characterization techniques, as well as the benefits and exclusive modes that the Asylum Research Cypher™ and MFP-3D™ AFMs offer. Researchers will learn more about evaluating local electrical properties, including current, surface charge and potential, dielectric breakdown, conductivity, and permittivity.
The application note can be downloaded at www.oxford-instruments.com/electrical-characterization.
Not only do the dimensions of silicon-based devices keep shrinking to a few nanometers, but also next- generation processes with nanoscale components like nanotubes, graphene, and molecular building blocks are emerging. Understanding physical processes that control electrical behavior increasingly
requires AFM measurements on smaller length scales,” said Keith Jones, Asylum Research Applications Scientist, specializing in electrical characterization. “This application note is a great reference for scientists new to AFM as well as those currently working in the field.”
Asylum Research AFMs are being used by leading researchers around the globe for characterizing nanoelectrical properties. A variety of their publications can be found at: www.oxford-instruments.com/nanoelectrical-afm.
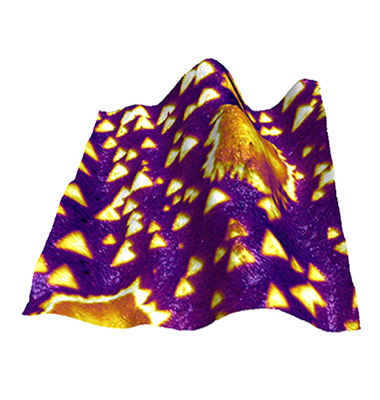
Figure caption: Kelvin Probe Force Microscopy surface potential overlaid on topography for flakes of boron nitride (small triangles) and graphene (large irregular features) grown on a copper foil substrate.
About Oxford Instruments Asylum Research
Oxford Instruments Asylum Research is the technology leader in atomic force microscopy for both materials and bioscience research. Asylum Research AFMs are widely used by both academic and industrial researchers for characterizing samples from diverse fields spanning material science, polymers, thin films, energy research, and biophysics. In addition to routine imaging of sample topography and roughness, Asylum Research AFMs also offer unmatched resolution and quantitative measurement capability for nanoelectrical, nanomechanical and electromechanical characterization. Recent advances have made these measurements far simpler and more automated for increased consistency and productivity. Its Cypher™ and MFP-3D™ AFM product lines span a wide range of performance and budgets. Asylum Research also offers its exclusive SurfRider™ AFM probes among a comprehensive selection of AFM probes, accessories, and consumables. Sales, applications and service offices are located in the United States, Germany, United Kingdom, Japan, France, India, China and Taiwan, with distributor offices in other global regions.
About Oxford Instruments plc
Oxford Instruments designs, supplies and supports high-technology tools and systems with a focus on research and industrial applications. Innovation has been the driving force behind Oxford Instruments’ growth and success for over 50 years, and its strategy is to effect the successful commercialisation of these ideas by bringing them to market in a timely and customer-focused fashion.
The first technology business to be spun out from Oxford University, Oxford Instruments objective is to be the leading provider of new generation tools and systems for the research and industrial sectors with a focus on nanotechnology. Its key market sectors include nano-fabrication and nano-materials. The company’s strategy is to expand the business into the life sciences arena, where nanotechnology and biotechnology intersect.
This involves the combination of core technologies in areas such as low temperature, high magnetic field and ultra high vacuum environments; Nuclear Magnetic Resonance; x-ray, electron, laser and optical based metrology; atomic force microscopy; optical imaging; advanced growth, deposition and etching.
Oxford Instruments aims to pursue responsible development and deeper understanding of our world through science and technology. Its products, expertise, and ideas address global issues such as energy, environment, security and health.




 Topics Increase Your AFM Knowledge
Topics Increase Your AFM Knowledge